 They are brittle in the solid state and are relatively good conductors of electricity. Metalloids exhibit characteristics of both metals and nonmetals. Nonmetals also have high electronegativity. Nonmetals are poor conductors of heat and electricity, dull and brittle. The physical characteristics of a metal are malleable, ductile, and shiny. A metal is an element, compound, or alloy that conducts electricity and heat. 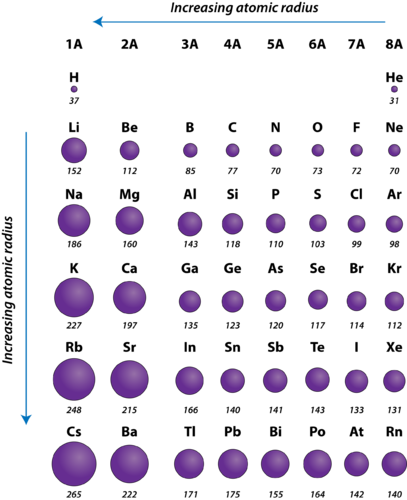 
Electron affinity decreases going down the groups and increases to the right.ĩ.) What are the characteristics of a metal, a nonmetal, a metalloid, and a noble gas? Electron affinities of the elements in period 3 and group 1 are similar to the trends in ionization energies. The line is not as smooth as you move across the row because it take more energy to remove an out-shell electron than an inner-shell electron.Ĩ.) How would the trend in electron affinities of the elements in period 3 and group 1 compare to the results you observed for ionization energies? Explain. Make sure you explain why the line is not smooth as you move across the row. Generally, the first ionization increases from left to right from the graph.ħ.) Explain the factors that determine these trends in ionization energies. According to the graph, as you move down a group the first ionization energy decreases because the valence electrons are farther away from the nucleus and have a weaker attraction to the positive nucleus. The first ionization energy is the energy required to remove the first electron from gaseous atoms or ions.Ħ.) Use your graphs to describe the trends in ionization energies as you move across the row and down the group However, if the atom has a negative charge, then the addition of electrons will cause a shielding effect and electron repulsion, so the ionic radius will be larger than the atomic radius. If an electron is taken away there will be less electron repulsion, therefore, the ionic radius will be smaller. If the charge is positive, the ionic radius would be less than the atomic radius because an electron is being taken away. The trends for the ionic radii of period 3 and group 1 depend on the ion’s charge. The ionic radius is the radius of an atom’s ion. The shielding effect is when the attraction between electrons and the positive nucleus decrease in any atom with the additional energy levels.Ĥ.) How would the trend in ionic radii of period 3 and group 1 compare to the results you observed for atomic radii? Explain. Each additional energy level causes the shielding effect to occur. The atomic radius increases from top to bottom because an additional energy level is added each row. The atomic radius decreases from left to right because the positive attraction of the electron due to the increasing number of protons. 
The graphsģ.) Explain the factors that determine these trends in atomic radius 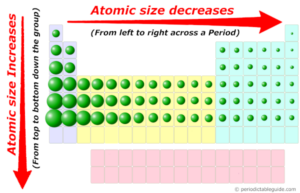
According to the graph, the atomic radius increase form top to bottom and decreases from left to right. Because electron cloud is a probability, in order to find the atomic radius, find the distance between the nucleuses of two atoms and divide it by 2.Ģ.) Use your graphs to describe the trends in atomic radius as you move across the row and down the group The atomic radius is the distance from the electron cloud to the nucleus. The atomic radius is used to describe the size of an atom. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
